Safety does not depend on geography alone. IVF abroad is safe when performed in well-regulated, experienced IVF clinics with strong laboratory standards and proper patient screening.
IVF safety abroad
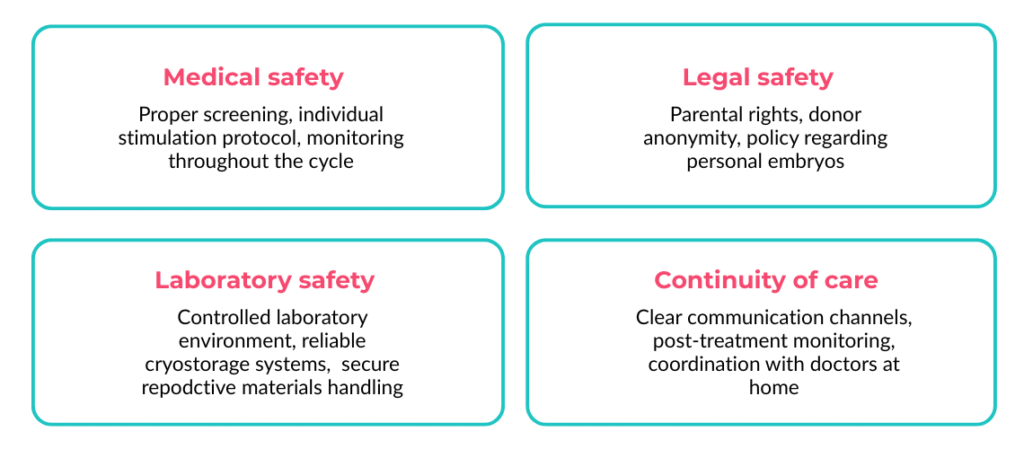
- Safety of IVF abroad has four dimensions: medical, laboratory, legal, and continuity of care
- IVF abroad is safe when performed in regulated, experienced clinics — not because of the country, but because of the standards
- Fertility clinic’s standards matter more than country branding
- Donor anonymity, age eligibility, and parental rights follow the clinic’s country law — not the patient’s home country law
- An affordable clinic with strong lab standards is safer than an expensive clinic with weak embryology protocols.
- A clinic that accepts patients without full screening isn’t cutting corners — it’s creating risk
- Lab quality decides embryo outcomes. Everything else is secondary
- The right IVF clinic abroad is the one matched to your age, diagnosis, and medical history — not the one with the lowest price or the best brochure
What does “safe” mean in IVF treatment?
IVF safety is not a geography question. It comes down to verifiable factors such as:
- Legal and regulatory oversight
- Medical assessment and eligibility screening
- Laboratory standards and embryo handling
- Donors screening protocols
- Follow-up care coordination
Patients who evaluate these elements carefully can access safe, effective, and often more affordable fertility treatment abroad.
Professor Alan Thornhill, Fertility Expert and Coach at The Fertility Guy, offers an important frame for understanding why: “The fertility journey creates a lot of stress, vulnerability, and a loss of control, making trust even more important than in simpler transactions. The treatment is financially, physically, and emotionally demanding, putting you at a disadvantage in many situations.” Safety, in his view, is inseparable from honesty and transparency — not just what happens in the laboratory, but how a clinic communicates at every stage.
How medically safe Is IVF abroad?
In 2022, over 960,000 ART treatment cycles were reported by 1,371 clinics across 39 European countries, according to ESHRE’s European IVF Monitoring (EIM) Consortium. Since 1997, EIM has recorded nearly 14 million treatments, resulting in 2.8 million children born across Europe.
Medical safety in IVF is a process, not a single event. It begins with:
- Comprehensive health assessment
- Individualized stimulation protocols
- Active cycle monitoring
- Age-appropriate health checks
Before evaluating any clinic abroad, it helps to understand which IVF risks exist everywhere — at home and abroad alike. OHSS, implantation failure, and multiple pregnancy are procedure-level risks tied to how IVF works biologically, not to where it is performed. A clinic in Spain carries the same baseline procedural risks as one in the UK. What differs between clinics is how well those risks are identified, managed, and minimised.
Laboratory safety: The factor that determines IVF outcomes more than any other
A clinic’s reputation is visible. Its embryology laboratory is not — but it determines more about your outcome than any other single factor. Lab quality directly affects embryo survival through vitrification, fertilisation rates, and blastocyst development.
Every clinic you evaluate, regardless of country or price, should be able to answer these questions clearly:
- Does the laboratory hold independent accreditation — ISO certification or ESHRE membership?
- Is there a documented, auditable chain of custody for every embryo from fertilisation to transfer?
- How is the laboratory environment monitored continuously — not just during working hours?
- What are the cryobank storage protocols in the event of a power failure?
- Who is responsible if a traceability failure occurs — and what is the documented protocol?
Dr. Claudia Mika, Founder and CEO of Temos International Healthcare Accreditation, explains what third-party accreditation actually requires: clinics must follow external standards defined by an international accreditation body, review their processes, update documentation, measure and record outcomes, and document incidents of malpractice. “In the end,” she says, “it’s to ensure that the treatment takes place in a safe, secure, trustworthy, and third-party accredited environment — and that the continuum of care is there from the first contact to the follow-up.”
Legal & ethical safety: Your home country’s law stops at the border
Most patients research clinic success rates before they research clinic legal frameworks. That order should be reversed. The moment you sign a consent form abroad, the law governing your donor’s anonymity, your parental rights, and the fate of any unused embryos is the law of the country you are sitting in — not the one you flew from.
A legally safe IVF clinic operates within a documented framework covering non-negotiable areas:
- Donor regulations — clear legal rules governing who can donate, under what conditions, and with what rights
- Consent procedures — fully documented, informed consent obtained from all parties before any procedure begins
- Embryo transfer policies — defined limits on the number of embryos transferred per cycle, backed by regulatory oversight
- Eligibility rules — regulated criteria determining who qualifies for treatment, including age limits and family structure
JCI, ISO, ESHRE: What these accreditations mean for your IVF safety
An accreditation badge tells you a process was audited — it does not tell you which process, or how recently. JCI certification evaluates hospital-wide patient safety systems, not embryology-specific protocols. ISO 9001 verifies quality management processes; ISO 15189 is the more relevant standard for medical laboratories. ESHRE membership reflects professional affiliation and data transparency — valuable, but not equivalent to regulatory oversight. Read every accreditation by asking three questions: what exactly was assessed, when was the last audit, and does it apply to the embryology laboratory specifically? A clinic that cannot answer all three is not fully accredited in any meaningful sense.
Accreditation standards are specific about who can work in the laboratory. Dr. Mika notes that embryologists in accredited organisations must be fully trained in all aspects of assisted reproductive technology: the head of the laboratory must hold an academic degree, laboratory supervisors must have at least three years of human embryology experience, and clinical embryologists must hold relevant biomedical qualifications. “It’s clearly stated what kind of background, quality and experience they have to have,” she explains.
Continuity of care: The part of IVF abroad that most clinics underpromise and most patients undercheck
Patients research stimulation protocols, laboratory accreditation, and success rates. Few research what happens when they land back home and something goes wrong. Continuity of care — the documented coordination between your foreign clinic and your local doctor — is the most commonly overlooked dimension of IVF safety abroad, and the one most likely to determine your experience if complications arise.
A safe clinic abroad documents its aftercare framework before treatment begins — not after complications arise. That framework must cover three areas:
- Clear communication channels — a named clinical contact, defined response times, and a documented protocol for urgent queries across time zones
- Post-treatment monitoring — a structured schedule for blood tests, ultrasound, and pregnancy confirmation, coordinated in your home country
- Coordination with your local doctor — written clinical summaries shared with your home physician before you leave,
A clinic may be affordable and well-known but unsafe if one of these dimensions is weak.
Key safety considerations for IVF abroad
Most patients begin their search for an IVF clinic abroad with two questions: how much does it cost, and what are the success rates? Both matter — but neither tells you whether the clinic is safe. The considerations that determine real safety are less visible, more specific, and almost never covered in a clinic brochure.
| Country | Description | Regulatory base |
| Spain Explore IVF Clinics in Spain | Known for strict regulation, advanced laboratories, and strong donor programs | Strict (SEF+EU-aligned) |
| Czech Republic Compare IVF Clinics in the Czech Republic | Combines affordability with modern fertility clinics and international patient experience | Strict (EU-aligned) |
| Greece Check IVF Clinics in Greece | Offers broad treatment options and experienced fertility specialists. | Strict (EU-aligned) |
| North Cyprus Compare IVF Clinics in North Cyprus | Known for flexible regulations and access to advanced fertility procedures. | Flexible — less formally structured regulatory environment |
| Portugal Explore IVF Clinics in Portugal | Growing in popularity IVF destination with advanced laboratory techniques. | Strong — nationally enforced, EU-aligned |
A strictly regulated destination does not guarantee a safe clinic. A flexible regulatory environment does not make one unsafe. What matters is knowing exactly which legal framework governs your treatment — and what it does and does not protect.
Professor Thornhill makes the link between legal compliance and patient safety explicit: “Compliance with local laws and regulations ensures safety, honesty, transparency, and a level playing field. Non-compliance can lead to legal risks and potential issues in your treatment cycle. Compliance indicates a commitment to honesty and transparency in clinic operations.”
Struggling to find an IVF clinic abroad? Connect with our Patient Care Advisor today!
Travel safety in IVF abroad: The risks are not in the flight — they are in the planning gap
Flying to Spain or Greece for IVF does not make the treatment riskier. Flying back without a documented aftercare plan does. The logistics that determine travel safety in IVF are not about the journey — they are about what is confirmed before it begins:
- Duration of stay — an incomplete cycle caused by early departure is a medical risk, not a scheduling inconvenience
- Recovery time after procedure
- Language support — informed consent is only legally and ethically valid when the patient fully understands what they are signing
- Home doctor coordination — a foreign clinic that does not proactively share your clinical summary with your local physician is not providing complete care
- Post-transfer monitoring — the two-week wait is not passive; it requires scheduled blood tests and ultrasound
Data privacy is a dimension of safety that patients rarely think to ask about. Sergio Gonzalez of Fenomatch, which provides donor-matching technology used by fertility clinics internationally, explains the baseline: “The medical data of a patient is sensitive data and has the highest level of protection. In European countries there is GDPR; for the US it is HIPAA. So no matter if you are a clinic, gamete bank, or a company, you must obey those regulations.” Patients treating abroad should confirm that any clinic handling their personal health data, genetic information, and medical history operates under one of these frameworks or an equivalent national standard.
Experienced fertility clinics often minimize travel by organizing testing before arrival.
Building a safe travel protocol around your IVF cycle
A safe IVF travel protocol is not built at the airport — it is built weeks before departure. Agree a clear communication protocol with your clinic — who you call, how quickly they respond, and what happens if your cycle runs longer than planned. Finally, build a contingency into every booking: flexible flights, cancellable accommodation, and a home doctor who has your treatment summary before you board. If something changes mid-cycle — and it sometimes does — a patient with a plan is in a fundamentally different position to one without.
Patient experiences with IVF abroad — what real outcomes look like
The Fertility Road Fertility Journey Project has followed real IVF patients from their first consultation through to final outcome since 2014, documenting what the process genuinely involves. The stories cover clinics across Spain, Greece, the Czech Republic, Cyprus, and Scandinavia.
Nathalie, a solo patient from France, gave birth to baby Charlie following double donation treatment at UR Vistahermosa in Spain— read the story UR Vistahermosa Alicante: Nathalie’s Solo Journey to Motherhood Through Double Donation
Laura and Dan from Exeter welcomed their son Charlie— see if your story is similar to Dreams Come True: Laura and Dan Welcome Their Beautiful Baby Charlie
What separates the experiences that patients describe as safe and well-managed from those they describe as stressful and disorganised is rarely the clinical outcome — it is the quality of communication, the clarity of the aftercare plan, and whether the patient felt genuinely informed at every stage of the process.

Safe IVF abroad checklist: Seven things to verify before you commit to any clinic
Choosing a clinic abroad safely is not a single decision — it is a structured verification process. A clinic that cannot provide clear answers to every item on this list is not ready to treat you safely. Work through each point before signing any consent form or making any payment.
- Legal framework — confirm which national law governs your treatment, donor rules, parental rights, and embryo ownership before anything else
- Full medical screening — hormonal profiling, ovarian reserve assessment, infectious disease testing, and full medical history required before your protocol is designed
- Accredited laboratory — independent certification of the embryology lab itself, not just the wider clinic or hospital
- Transparent donor program — documented screening protocols, clear anonymity or identity-release policy, and legally defined compensation structure
- Cost breakdown — itemised pricing covering all cycle stages, medication, monitoring, and cryostorage — with no significant costs disclosed only after commitment.
- Treatment timeline — a written protocol with confirmed dates, stay duration, and a defined procedure for cycle extensions or delays
- Post-treatment coordination — a documented aftercare plan including home monitoring schedule, named clinical contact, and written handover to your local doctor
The safest choice is not necessarily the cheapest. It is the one aligned with your medical profile and long-term health.
Prof. Thornhill frames cost transparency as a patient safety issue, not just a financial one: “Hidden, unexpected or extra costs can cause shock and stress, and sometimes even result in the stopping of treatment.” His practical advice: always request a written itemised plan before committing.
Conclusion
The safest IVF abroad choice aligns with your medical profile, not the lowest price tag.
FAQ
Is IVF abroad riskier than IVF at home?
Not necessarily. Risks depend on IVF clinic quality, medical protocols, and patient health not the IVF destination alone.
Can women over 50 have IVF abroad?
Age limits vary by country and fertility clinic. Eligibility must be confirmed in advance.
Is donor egg IVF safe abroad?
Yes, when egg donor screening, legal protections, and medical protocols are properly structured.
Struggling to find an IVF clinic abroad? Connect with our Patient Care Advisor today!
Updated: 13.04.2026